A) NH3
B) Ca(OH) 2
C) Al(OH) 3
D) B(OH) 3
E) CH3OH
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 3.682-g sample of KClO3 is dissolved in enough water to give 375.mL of solution.What is the chlorate ion concentration in this solution?
A) 9.82 M
B) 1.60 × 10-1 M
C) 8.01 ×10-2 M
D) 9.82 × 10-3 M
E) 1.60 × 10-2 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is false concerning oxidation numbers?
A) An element's oxidation number is constant and unchanging.
B) Atoms of free elements have an oxidation number of zero.
C) An atom's oxidation state signifies the number of charges for that atom in a molecule.
D) Oxidation numbers allow easy identification of which atoms gained or lost electrons.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which salt is produced by the neutralization of sodium hydroxide with sulfuric acid?
A) Na2SO4
B) Na2(SO4) 3
C) Na(SO4) 2
D) Na2SO3
E) Na2S
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Which represents the result of mixing equal volumes of 1 M HCl and 1 M NH3 solutions? (Each sphere represents 1 mol of ions.)
A. 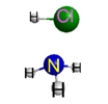 B.
B. 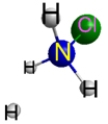 C.
C. 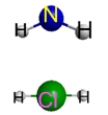 D.
D. 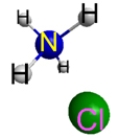 E.
E. 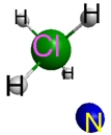
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of iodine in I2?
A) -1
B) 0
C) +1
D) +7
E) -7
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of Cr in Cr2O72-?
A) -12
B) -7
C) -2
D) +6
E) +7
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following oxidation-reduction equation: Al(s) + Ag+(aq) → Al3+(aq) + Ag(s)
A) Al(s) + Ag+(aq) → Al3+(aq) + Ag(s)
B) 3Al(s) + Ag+(aq) → 3Al3+(aq) + Ag(s)
C) Al(s) + 3Ag+(aq) → Al3+(aq) + 3Ag(s)
D) 3Al(s) + Ag+(aq) → Al3+(aq) + 3Ag(s)
E) Al(s) + 3Ag+(aq) → 3Al3+(aq) + Ag(s)
G) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which substance is present in the smallest proportion in a solution?
A) liquid
B) gas
C) solvent
D) solid
E) solute
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
H3PO4 is a strong acid.
B) False
Correct Answer

verified
False
Correct Answer
verified
Short Answer
In each of the following cases,write down the oxidation number of the indicated atom. a.P in P4 b.C in C2H6 c.S in H2SO4 d.Mn in MnO4- e.P in Na3PO4
Correct Answer

verified
a = 0,b = ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which element is reduced in the following reaction? Cu + 2H2SO4 → CuSO4 + SO2 + 2H2O
A) Cu
B) H
C) S
D) O
E) H2O
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a weak base?
A) NH3
B) Ca(OH) 2
C) Ba(OH) 2
D) NaOH
E) CH3COOH
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n)___________ ____________ is oxidized during a redox reaction and causes another substance to undergo reduction.
Correct Answer

verified
Correct Answer
verified
Short Answer
___________ is a concentration unit that is defined as the number of moles of solute per liters of solution.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sodium ions are present in 325 mL of 0.850 M Na2SO4?
A) 5.12 × 1023 Na ions
B) 8.32 × 1022 Na ions
C) 1.02 × 1024 Na ions
D) 1.66 × 1023 Na ions
E) 3.33 × 1023 Na ions
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the net ionic equation for the following reaction: 2NaCl(aq) + Hg2(NO3) 2(aq) → 2NaNO3(aq) + Hg2Cl2(s)
A) Na+(aq) + NO3- (aq) → NaNO3(aq)
B) Hg22+(aq) + 2Cl-(aq) → Hg2Cl2(s)
C) 2Hg+(aq) + 2Cl-(aq) → 2HgCl(s)
D) Hg2(NO3) 2(aq) → Hg22+(aq) + 2NO3-(aq)
E) Hg22+(aq) → Hg2(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is reduced in the following reaction? Cu(NO3) 2 + Zn → Zn(NO3) 2 + Cu
A) Cu(NO3) 2
B) N
C) O
D) Zn
E) NO3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a weak acid?
A) H2SO4
B) HNO3
C) HF
D) HBr
E) HCl
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the reducing agent in the following reaction? 5Fe2+(aq) + MnO4-(aq) + 8H+(aq) → 5Fe3+(aq) + Mn2+(aq) + 4H2O(l)
A) Fe2+
B) MnO4-
C) H+
D) Mn2+
E) Fe3+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 144
Related Exams