A) Beta decay
B) Fission reaction
C) Fusion reaction
D) Alpha decay
E) Positron emission
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are magic numbers with reference to nuclear stability?
A) The number of nuclear particles of elements not located in the belt of stability
B) The number of nuclear particles of radioactive elements
C) The number of nuclear particles in generally more stable nuclei
D) None of the
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
It is believed that two carbon-12 nuclei can react in the core of a supergiant star to form sodium-23 and hydrogen-1.Calculate the energy released from this reaction for each mole of hydrogen formed. 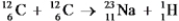 Particle Mass (amu) C-12 12.000000
Na-23 22.989767
H-1 1.007825
(1 kg = 6.022 × 1026 amu;
NA = 6.022 × 1023 mol-1;
C = 2.99792458 × 108 m/s)
Particle Mass (amu) C-12 12.000000
Na-23 22.989767
H-1 1.007825
(1 kg = 6.022 × 1026 amu;
NA = 6.022 × 1023 mol-1;
C = 2.99792458 × 108 m/s)
A) 2.16 × 1014 kJ
B) 2.16 × 1011 kJ
C) 2.16 × 108 kJ
D) 2.16 × 105 kJ
E) None of the answers is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following type(s) of radiation is/are detected by a Geiger counter?
A) Beta particles
B) Alpha particles
C) Gamma rays
D) Beta particles and gamma rays
E) Alpha particles,beta particles,and gamma rays
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Gamma rays are not deflected by an electric field.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose you have two very sensitive balances that can measure the masses of individual particles,depicted below.In these figures,"p" corresponds to a proton,and "n" corresponds to a neutron. 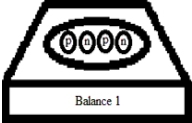
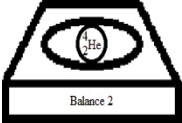 What is the relationship between the mass readings of the two balances? (Neglect differences due to experimental uncertainty.)
What is the relationship between the mass readings of the two balances? (Neglect differences due to experimental uncertainty.)
A) The mass reading of balance 1 will be much higher than the mass reading of balance 2.
B) The mass reading of balance 1 will be slightly higher than the mass reading of balance 2.
C) The mass reading of balance 1 will be exactly the same as the mass reading of balance 2.
D) The mass reading of balance 1 will be slightly lower than the mass reading of balance 2.
E) The mass reading of balance 1 will be much lower than the mass reading of balance 2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nuclear process called in which a heavy nuclear of mass number greater than 200 divides to form smaller nuclei of intermediate mass and one or more neutron(s) ?
A) Photonuclear reactions
B) Nuclear fission
C) Thermal conductivity
D) Nuclear combination
E) Nuclear fusion
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What technique would you use to determine how old a cloth is that was found while digging in Egypt?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is injected into the bloodstream to trace the flow of blood and detect possible constrictions or obstructions in the circulatory system?
A) (18O)
B) (131I)
C) (123I)
D) (24Na)
E) (99Tc)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
When a87 Br nucleus emits a beta particle,what is the nuclear species that results?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An isotope with a high value of N/Z will tend to decay through
A) α decay.
B) β decay.
C) positron decay.
D) electron capture.
E) γ decay.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An 85-kg person exposed to barium-141 receives 2.5 × 105 β particles,each with an energy of 5.2 × 10-13 J.How many rads does the person receive? (1 rad = 1 × 10-5 J/g)
A) 2.4 × 10-20 rads
B) 1.5 × 10-7 rads
C) 1.8 × 10-16 rads
D) 6.1 × 10-15 rads
E) None of the answers is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following best represents the relationship between the energy released (ΔE) and the mass defect (Δm) ?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following materials is put into a nuclear reactor to slow the chain reaction?
A) Heavy water
B) Moderators
C) Control rods
D) Reflectors
E) Chlorine
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When atoms of aluminum-27 are bombarded with alpha particles,a neutron and an element are produced.Which particular isotope of this element is formed? 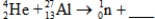
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the nuclear process that results from the bombardment of nuclei by neutrons,protons,or other nuclei?
A) Nuclear transmutation
B) Protonation
C) Nucleation
D) Nuclear condensation
E) Radioactivity
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A plot of the number of neutrons versus the number of protons in various isotopes produces a "belt of stability." Isotopes below the belt of stability (i.e.,with a smaller neutron-to-proton ratio)decay by beta particle emission.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Petroleum is a fossil fuel containing many different carbon compounds.If the carbon atoms in petroleum have been in the ground for 100 million years,what fraction of the initial 14C atoms is still there? (t1/2 = 5730 yr)
A) 0
B) 1 × 10-10
C) 5.7 × 10-5
D) 1.0 × 10-3
E) 5.7 × 10-1
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium-21 will emit positrons,each having an energy of 4.0 × 10-13 J.What is this energy in MeV? (1 MeV = 1.602 × 10-13 J)
A) 4.0 × 10-7 MeV
B) 2.5 MeV
C) 40 MeV
D) 2.5 × 106 MeV
E) 2.5 × 10-6 MeV
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The _______________ has the same mass as the electron,but bears a charge of +1.
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 125
Related Exams