Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a liquid component of gasoline. Given the following vapor pressures of octane at various temperatures, estimate the boiling point of octane in Leadville, Colorado, where the atmospheric pressure is 496 mmHg. 400 mmHg @ 104°C, 500 mmHg @ 111°C, 600 mmHg @ 117°C, 700 mmHg @ 122°C, 760 mmHg @ 125°C
A) 125°C
B) 120°C
C) 115°C
D) 110°C
E) 105°C
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following compound and its boiling point, identify whether it is polar or nonpolar: F2, -188.1°C.
Correct Answer

verified
Correct Answer
verified
Short Answer
Which is expected to have a higher boiling point, C8H18 or C4H10?
Correct Answer

verified
Correct Answer
verified
Short Answer
Boron nitride, BN3, melts at approximately at 3,000°C under high pressure.This materal is almost as hard as diamond.What kind of crystal is this?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Krypton has a higher melting point than argon because of its
A) hydrogen bonding.
B) stronger dispersion forces.
C) permanent dipole moment.
D) ionic bonds.
E) greater ionization energy.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in H2S(g).
Correct Answer

verified
Correct Answer
verified
Short Answer
What phase exists at the point labeled c? 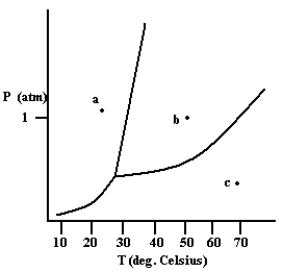
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in CHCl3(l).
Correct Answer

verified
dipole-dip...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following properties indicates the presence of strong intermolecular forces in a liquid?
A) a low heat of vaporization
B) a low critical temperature
C) a low vapor pressure
D) a low boiling point
E) None of the above.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the amount of heat needed to melt 2.00 kg of iron at its melting point (1,809 K) , given that Hfus = 13.80 kJ/mol.
A) 494 kJ
B) 27,600 kJ
C) 27.6 kJ
D) 27,600 J
E) 25,000 kJ
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in SF6(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A liquid boils when its
A) vapor pressure is exactly 1 atmosphere.
B) vapor pressure is equal to, or greater than, the external pressure pushing on it.
C) temperature is equal to 273 K (standard temperature) .
D) temperature is greater than room temperature.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be expected to have the lowest vapor pressure at room temperature?
A) ethanol, bp = 78°C
B) methanol, bp = 65°C
C) water, bp = 100°C
D) acetone, bp = 56°C
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the following species are the dispersion forces strongest?
A) C4H10
B) C5H12
C) C6H14
D) C7H16
E) C8H18
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances crystallizes as a molecular solid?
A) KI
B) SiO2
C) Sn
D) CH3OH
E) Al2(SO4) 3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the temperature at which ethanol boils on a day in the mountains when the barometric pressure is 547 mmHg.(Given: The heat of vaporization of ethanol is 39.3 kJ/mol; the normal boiling point of ethanol is 78.3°C.)
A) 76.5°C
B) 69.9°C
C) 10.0°C
D) 77.9°C
E) 74.6°C
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic planes in a graphite crystal are separated by 335 pm. At what angle would you find the first-order (n = 1) diffraction of 0.154 nm X-rays from a graphite crystal?
A) 27.4°
B) 66.8°
C) 0.232°
D) 13.3°
E) 2.63°
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances is expected to have the highest boiling point?
A) Br2
B) Cl2
C) F2
D) I2
F) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in HCl(g).
Correct Answer

verified
dipole-dip...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 101 - 120 of 140
Related Exams