A) 0.025
B) 0.94
C) 1.60
D) 12.40
E) 10.31
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate acid of CO32− in the reaction below? CO32− + HSO4−  HCO3− + SO42−
HCO3− + SO42−
A) HCO3−
B) HSO4−
C) OH−
D) H3O+
E) SO42−
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A standard solution of 0.243 M NaOH was used to determine the concentration of a hydrochloric acid solution.If 46.33 mL of NaOH is needed to neutralize 10.00 mL of the acid, what is the molar concentration of the acid in the original solution?
A) 0.243 M
B) 0.200 M
C) 1.13 M
D) 0.563 M
E) 0.0313 M
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these salts will form a basic solution upon dissolving in water?
A) NaCl
B) NaNO2
C) NH4NO3
D) KBr
E) AlCl3
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 34.62-mL of 0.1510 M NaOH was needed to neutralize 50.0 mL of an H2SO4 solution.What is the concentration of the original sulfuric acid solution?
A) 0.0229 M
B) 0.218 M
C) 0.0523 M
D) 0.209 M
E) 0.105 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Vinegar is a solution of acetic acid, HC2H3O2 , dissolved in water.A 5.54-g sample of vinegar was neutralized by 30.10 mL of 0.100 M NaOH.What is the percent by weight of acetic acid in the vinegar?
A) 0.0543%
B) 1.08%
C) 3.26%
D) 5.43%
E) 9.23%
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these responses is with regard to a 0.1 M solution of a weak acid HA?
A) [H+] > 0.1 M.
B) pH = 1.0
C) [H+] < [A-]
D) pH > 1.0
E) [OH-] > [H+]
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of a proton acceptor in an acid-base reaction?
A) Arrhenius acid
B) Arrhenius base
C) Brønsted acid
D) Brønsted base
E) Lewis base
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following reaction and identify the Brønsted base: NaOH(aq) + HCl(aq) →
A) Na(aq) + HOCl(aq) ; NaOH is the base.
B) NaCl(aq) + OH−(aq) ; HCl is the base.
C) NaCl(aq) + OH−(aq) ; NaOH is the base.
D) H2O(l) + NaCl(aq) ; HCl is the base.
E) NaCl(aq) + H2O(l) ; NaOH is the base.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to a substance that can act as a Brønsted acid or as a Brønsted base according to what it is reacting with?
A) hydrophilic
B) hydrophobic
C) amphoteric
D) isoprotic
E) isoelectronic
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which pair of substances is capable of forming a buffer in aqueous solution?
A) H3PO4, Na3PO3
B) HNO3, NaNO3
C) HCl, NaCl
D) H2CO3, NaNO2
E) CH3COOH, CH3COONa
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the concentration of H+ in a 0.025 M HCl solution?
A) 0
B) 0.013 M
C) 0.025 M
D) 0.050 M
E) 0.010 M
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution in which [H+] is 1.22 × 10−4 M?
A) −3.914
B) 1.220
C) 3.914
D) 4.000
E) 4.086
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the reaction, HSO4
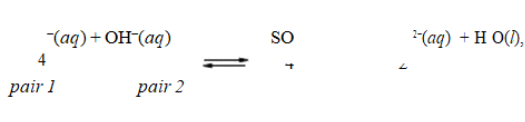 , the conjugate acid-base pairs are
, the conjugate acid-base pairs are
A) HSO − and SO 2−; H O and OH−.4 4
2 ![]()
B) HSO4− and H3O+; SO42 − and OH−.
C) HSO4− and OH−; SO42 − and H2O.
D) HSO4− and H2O; OH − and SO42−.
E) HSO4− and OH−; SO42 − and H3O+.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of a proton donor in an acid-base reaction?
A) Arrhenius acid
B) Arrhenius base
C) Brønsted acid
D) Brønsted base
E) Lewis base
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the concentration of OH− in a 0.083 M NaOH solution?
A) 0.083 M
B) 1.21 × 10−13 M
C) 8.30 × 10−16 M
D) 7 M
E) 8.30 × 1012 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the conjugate base of HPO42− in the reaction HCO3− + HPO42−  H2CO3 + PO43−
H2CO3 + PO43−
A) H2O
B) HCO3−
C) H2CO3
D) PO43−
E) None of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the net ionic equation for the reaction between aqueous solutions of lithium hydroxide and hydrobromic acid? LiOH(aq) + HBr(aq) → H2O(l) + LiBr(aq)
A) LiOH(aq) → Li+(aq) + OH−(aq)
B) HBr(aq) → H+(aq) + Br−(aq)
C) H+(aq) + OH−(aq) → H2O(l)
D) Li+(aq) + Br−(aq) → LiBr(aq)
E) Li+(aq) + OH−(aq) + H+(aq) + Br−(aq) → H2O(l) + LiBr(aq)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a strong acid?
A) CH3COOH
B) H2SO3
C) NH3
D) H3PO4
E) HClO3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the major ions present in an aqueous HNO3 solution.
A) HN2+, O2−
B) OH−, NO3−
C) OH−, NO+
D) H+, N3−, O2−
E) H+, NO3−
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 65
Related Exams