A) 1.0 * 10-13 m
B) 1.0 * 10-10 m
C) 1.0 * 10-7 m
D) 1.0 m
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The quantum numbers, n = 4, l = 1, ml = 1, ms = +1/2, represent an electron in a ______ subshell.
A) 4s
B) 3s
C) 4p
D) 3p
E) None of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of a ball bearing with a mass of 10.0 g, and a velocity of 10.0 cm/s
A) 4.63 * 10-22 nm
B) 5.63 * 10-22 nm
C) 6.63 * 10-22 nm
D) 7.63 * 10-22 nm
E) None of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the wavelength associated with a 20Ne+ ion moving at a velocity of 2.0 * 105 m/s. The atomic mass of 20Ne is 19.992 amu.
A) 1.7 * 10-40 m
B) 1.0 * 10-18 m
C) 1.0 * 10-16 m
D) 1.0 * 10-13 m
E) 9.7 * 1012 m
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electron configuration of an excited state of an iron atom
A) [Ar]4s23d7
B) [Ar]4s23d6
C) [Ar]4s23d8
D) [Ar]4s13d7
E) [Ar]4s13d5
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration [Xe]6s24f145d10
A) Lu
B) Cd
C) Hg
D) Lr
E) Au
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Each shell (principal energy level) of quantum number n contains n subshells.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the figure below, categorize electromagnetic radiation with a wavelength of 1.0 x 10-1 m. 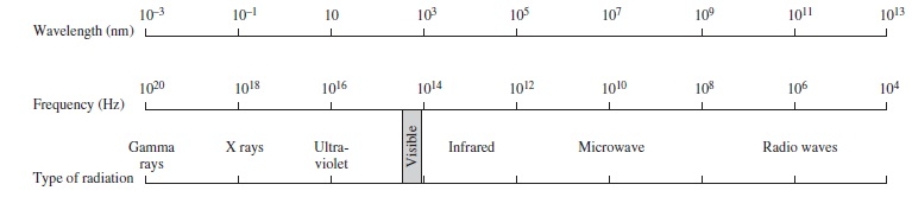
A) Gamma rays
B) X rays
C) Ultraviolet
D) Infrared
E) Microwave
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
An electron gains energy in the transition from a 4d subshell to a 5s subshell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons in an atom that can have the following set of quantum numbers N = 3 l = 1 ml = -1
A) 18
B) 6
C) 3
D) 2
E) 1
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms is diamagnetic in its ground-state
A) Ca
B) As
C) Cu
D) Fe
E) none of these
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the energy, in joules, required to excite a hydrogen atom by causing an electronic transition from the n = 1 to the n = 4 principal energy level. Recall that the energy levels of the H atom are given by En = -2.18 * 10-18 J(1/n2)
A) 2.07 * 10-29 J
B) 2.25 * 10-18 J
C) 2.04 * 10-18 J
D) 3.27 * 10-17 J
E) 2.19 * 105 J
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following frequencies of light has the highest energy
A) 2.5 x 1010 s-1
B) 7.0 x 1013 s-1
C) 2.3 x 1014 s-1
D) 5.0 x 1014 s-1
E) 1.4 x 1015 s-1
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which choice lists two elements with ground-state electron configurations that are well known exceptions to the Aufbau principle
A) Cu and C
B) Cr and Cu
C) Cs and Cl
D) Rb and Co
E) Fe and Co
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the figure below, categorize electromagnetic radiation with a wavelength of 1.0 x 10-3 m. 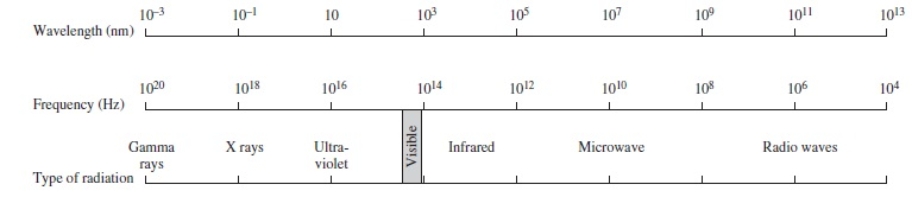
A) Gamma rays
B) X rays
C) Ultraviolet
D) Infrared
E) Microwave
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 135 of 135
Related Exams