A) Csp3 + Csp3, and C2p + C2p
B) Csp3 + Csp3, and Csp2 + Csp2
C) Csp2 + Csp2, and C2p + C2p
D) Csp2 + Csp2, and Csp2 + Csp2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pair does not represent resonance structures? 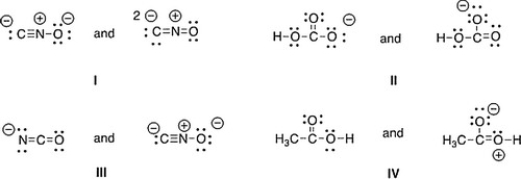
A) IV
B) II
C) III
D) I
F) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Avobenzone is an active ingredient in some common sunscreens. Which of the following is the correct molecular formula for avobenzone? 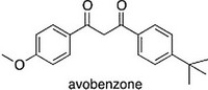
A) C20H24O3
B) C20H22O3
C) C21H23O3
D) C22O24O3
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the geometry around the indicated atom in each species. 
A) I = Trigonal planar; II = linear; III = tetrahedral; IV = trigonal planar
B) I = Tetrahedral; II = trigonal planar; III = linear; IV = tetrahedral
C) I = Linear; II = tetrahedral; III = trigonal planar; IV = tetrahedral
D) I = Linear; II = tetrahedral; III = trigonal planar; IV = linear
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a chlorine anion (Cl-) ?
A) 1s2, 2s2, 2p6
B) 1s2, 2s2, 2p6, 3s2, 3p5
C) 1s2, 2s2, 2p6, 3s2, 3p4
D) 1s2, 2s2, 2p6, 3s2, 3p6
F) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following molecules does not have a net dipole moment of zero?
A) BF3
B) CO2
C) NH3
D) CCl4
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is not true?
A) Resonance structures differ only in the arrangement of electrons.
B) There is no movement of electrons from one form to another.
C) Resonance structures are not isomers.
D) Resonance structures are in equilibrium with each other.
F) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is true?
A) Resonance structures have the same placement of atoms but different arrangement of electrons.
B) Resonance structures have the same placement of electrons but different arrangement of atoms.
C) Resonance structures have different placement of atoms and different arrangement of electrons.
D) Resonance structures have the same placement of atoms and the same arrangement of electrons.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has an atom with more than eight valence electrons?
A) H2CO3
B) H2SO4
C) HBr
D) H2O
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct Lewis structure for acetic acid (CH3CO2H) ? 
A) III
B) I
C) IV
D) II
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization for each of the indicated atoms in the following compound? 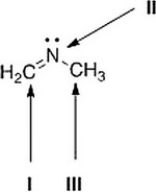
A) I = sp; II = sp2; III = sp3.
B) I = sp2; II = sp2; III = sp3.
C) I = sp2; II = sp3; III = sp3.
D) I = sp2; II = sp2; III = sp2.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not an acceptable Lewis structure for the anion CH2NCO-? 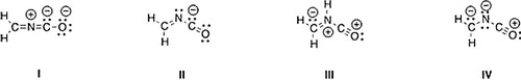
A) II
B) III
C) I
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements correctly describes the typical number of bonds for carbon, nitrogen, and oxygen in most neutral organic molecules?
A) Carbon forms 4 covalent bonds, nitrogen forms 3 covalent bonds, and oxygen forms 2 covalent bonds.
B) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds, and oxygen forms 4 covalent bonds.
C) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds, and oxygen forms 2 covalent bonds.
D) Carbon forms 4 covalent bonds, nitrogen forms 2 covalent bonds, and oxygen forms 3 covalent bonds.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 13 of 13
Related Exams