A) It is fastest with tertiary halides.
B) All bonds are broken and formed in a single step.
C) It exhibits first-order kinetics.
D) A better leaving group should make a faster reaction.
F) B) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 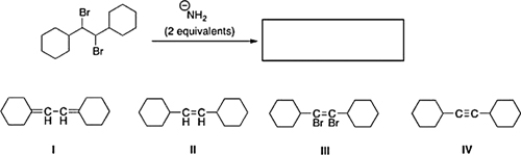
A) II
B) III
C) IV
D) I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Classify each alkene in vitamin D3 labeled I, II, III by the number of carbon substituents bonded to the double bond. 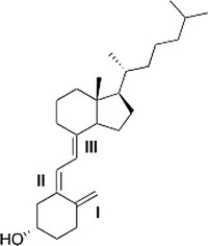
A) I = Monosubstituted; II = disubstituted; III = trisubstituted
B) I = Disubstituted; II = disubstituted; III = trisubstituted
C) I = Disubstituted; II = trisubstituted; III = disubstituted
D) I = Disubstituted; II = trisubstituted; III = trisubstituted
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Consider the following E2 reaction. What rate equation would be observed for this reaction? ![Consider the following E2 reaction. What rate equation would be observed for this reaction? A) Rate =k[CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>Br][KOC(CH<sub>3</sub>) <sub>3</sub>] B) Rate =k[CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>Br]<sup>2</sup>[KOC(CH<sub>3</sub>) <sub>3</sub>] C) Rate =k[CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>Br] D) Rate = k[CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>Br][KOC(CH<sub>3</sub>) <sub>3</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cf4_eadc_862d_49cab86c3690_TBMG1035_00.jpg)
A) Rate =k[CH3CH2CH2Br][KOC(CH3) 3]
B) Rate =k[CH3CH2CH2Br]2[KOC(CH3) 3]
C) Rate =k[CH3CH2CH2Br]
D) Rate = k[CH3CH2CH2Br][KOC(CH3) 3]2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 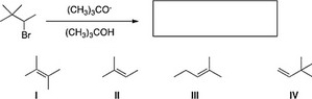
A) III
B) II
C) I
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about an E1 mechanism is true?
A) The reaction is slowest with tertiary substrates.
B) The reaction follows second-order kinetics.
C) The identity of the leaving group affects the rate of reaction.
D) Polar aprotic solvents favor the E1 mechanism.
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 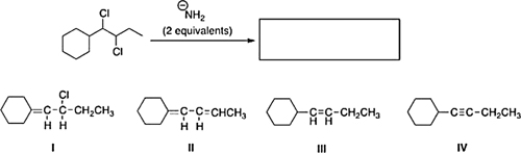
A) III
B) II
C) IV
D) I
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about an E1 mechanism isnot true?
A) The reaction follows first-order kinetics.
B) Stronger bases favor the E1 reaction.
C) The reaction is fastest with tertiary alkyl halides.
D) A better leaving group makes the reaction rate increase.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 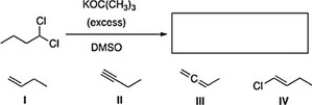
A) I
B) IV
C) III
D) II
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 9 of 9
Related Exams