Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for NOCl, a reactive material used as an ionizing solvent.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is most likely to be covalent?
A) CsOH
B) NF3
C) Sr(NO3) 2
D) CaO
E) LiF
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Describe in brief how electronegativity values can be used to predict the percent ionic character of a bond between two atoms.
Correct Answer

verified
Take the electronegativity difference (Δ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What types of elements undergo ionic bonding?
A) two metals
B) a nonmetal and a metal
C) two nonmetals
D) two Group 1A elements
E) two noble gases
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis structure for CS2 is:
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the compound with the lowest (i.e., least negative) lattice energy.
A) CsBr(s)
B) NaCl(s)
C) SrO(s)
D) CaO(s)
E) KBr(s)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Ionic compounds tend to form between metals and nonmetals when electrons are transferred from an element with high ionization energy (metal) to an element with a low electron affinity (nonmetal).
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many dots does the Lewis dot symbol for oxygen have around it?
A) 4
B) 2
C) 6
D) 3
E) 8
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on Cl in the structure shown for the perchlorate ion is ________. 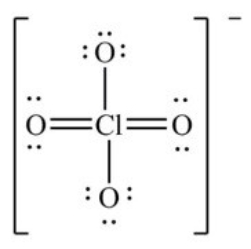
A) -2
B) -1
C) 0
D) +1
E) +2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A triple bond cannot exist between a carbon atom and a hydrogen atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many dots does the Lewis dot symbol for magnesium have around it?
A) 1
B) 2
C) 0
D) 3
E) 12
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Only valence electrons are shown in the Lewis structure held together by covalent bonds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is most likely to be ionic?
A) KF
B) CCl4
C) CS2
D) CO2
E) ICl
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ is a representation of covalent bonding in which shared electron pairs are shown either as dashes or as pairs of dots between two atoms and unshared electrons are shown as dots around the individual atoms.
Correct Answer

verified
Correct Answer
verified
True/False
A double bond cannot exist between a carbon atom and an oxygen atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these ionic solids would have the largest lattice energy?
A) NaCl
B) NaF
C) CaBr2
D) CsI
E) CaCl2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the electrostatic attraction called that holds oppositely charged ions together in a compound?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the compound with the highest (i.e., most negative) lattice energy.
A) CaS(s)
B) BaO(s)
C) NaI(s)
D) LiBr(s)
E) MgO(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many lone pairs of electrons need to be added to complete this Lewis structure? 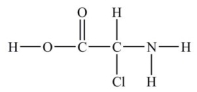
A) 5
B) 8
C) 6
D) 1
E) 16
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 102
Related Exams