Correct Answer

verified
Correct Answer
verified
True/False
Octahedral complexes can exhibit geometric and optical isomerism.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the label used for an isomer which is levorotatory?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following energy-level diagram could correspond to which coordination compound? 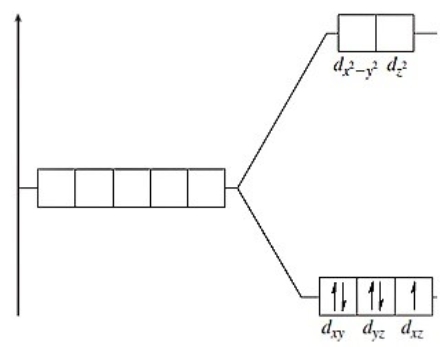
A) hexabromomanganate(II)
B) hexacyanoferrate(II)
C) hexacyanoferrate(III)
D) hexabromomanganate(IV)
E) hexachlorochromate(II)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Co(en) 2Cl2]Cl, the coordination number and oxidation number of the central atom are ________ and ________, respectively.
A) 4; +3
B) 6; +2
C) 4; +2
D) 6; +3
E) 4; +1
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
An equimolar mixture (50:50 ratio) of two enantiomers is called a(n) ________ ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron(III) forms an octahedral complex with the ligand CN-. How many unpaired electrons are in the d orbitals of iron?
A) 1
B) 3
C) 5
D) 7
E) 9
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal field splitting energy, Δ,
A) is larger for tetrahedral complexes than for octahedral complexes.
B) depends on the metal but not on the ligand.
C) determines the color of a complex.
D) is larger for ionic ligands like chloride than for molecular ligands like carbon monoxide, CO.
E) determines the charge of a complex.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the formula for diamminedichloroethylenediaminecobalt(III) bromide.
A) [CoCl2(en) (NH3) 2]Br
B) [CoCl2(en) (NH3) 2]Br2
C) [CoCl2(en) 2(NH3) 2]Br
D) [CoCl2(en) 2(NH3) 2]Br2
E) (NH3) 2Cl2(en) Co3Br
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these diagrams represent the crystal field splitting between d orbitals in a tetrahedral complex?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ion [Co(NH3) 6]2+ is octahedral and high spin. This complex is
A) paramagnetic, with 1 unpaired electron.
B) paramagnetic, with 3 unpaired electrons.
C) paramagnetic, with 4 unpaired electrons.
D) paramagnetic, with 5 unpaired electrons.
E) diamagnetic.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands has two different types of donor atoms?
A) NH3
B) H2O
C) NH4+
D) NO2-
E) ethylenediamine
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The maximum oxidation state of an element in the first transition series never exceeds its group number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the atom in a ligand that is bound directly to the metal atom?
A) Acceptor atom
B) Donor atom
C) Chelating atom
D) Coordinator
E) Chelating agent
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be paramagnetic?
A) V5+
B) Ni2+
C) Mn7+
D) Ti4+
E) Zn2+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a ground-state Sc3+ ion have?
A) 1
B) 3
C) 2
D) 10
E) 0
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Pt(NH3) 2Cl2], the coordination number and oxidation number of the central atom are ________ and ________, respectively.
A) 2; 0
B) 4; +4
C) 5; 0
D) 4; +2
E) 6; +2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many geometric isomers can the following square-planar complex have? 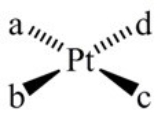
A) 1
B) 2
C) 3
D) 4
E) 6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In K4[Fe(CN) 6], how many 3d electrons does the iron atom have?
A) 3
B) 4
C) 5
D) 6
E) 7
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a ground-state Fe3+ ion have?
A) 3
B) 5
C) 6
D) 7
E) 9
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 132
Related Exams