A) II < III < IV < I
B) I < II < III < IV
C) IV < III < II < I
D) II < I < III < IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major organic product of the following reaction. 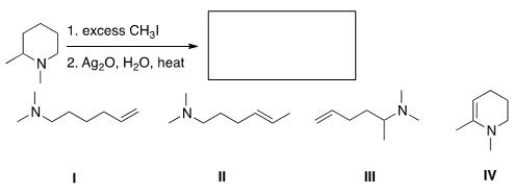
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is pyrrole much less basic than pyridine?
A) The lone pair of electrons in pyrrole is located on an sp2 orbital.
B) The lone pair of electrons in pyrrole is part of the aromatic p system.
C) The lone pair of electrons in pyrrole is not part of the aromatic p system.
D) The pKa of the conjugate acid of pyrrole is much greater than the conjugate acid of pyridine.
F) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Predict the product(s) of the following reaction. 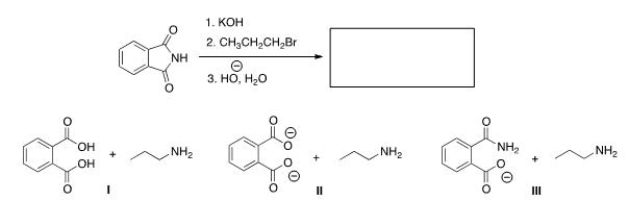
A) I
B) II
C) III
D) None of the choices
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct assignment of the names of the following aromatic amines?
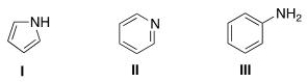
A) I = pyrrolidine; II = pyrimidine; III = aniline.
B) I = pyrrole; II = pyrimidine; III = anisole.
C) I = pyrrolidine; II = pyridine; III =aniline.
D) I = pyrrole; II = pyridine; III = aniline.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following amines are classified as tertiary (3°) amines?
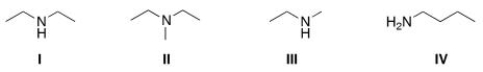
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
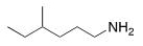
A) 3-methyl-1-hexanamine
B) 4-methyl-1-hexylamine
C) 4-methyl-1-hexanamine
D) 3-methyl-6-hexylamine
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major organic product of the following reaction. 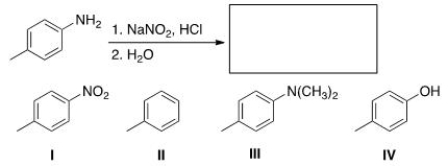
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the following compound?
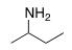
A) isopropylamine
B) sec-butylamine
C) isobutylamine
D) tert-butylamine
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the N-H bond of an imide especially acidic?
A) The conjugate base is stabilized by electron-donating inductive effect.
B) The conjugate base is stabilized by resonance.
C) The conjugate acid is stabilized by resonance.
D) The conjugate base is stabilized by intramolecular hydrogen bonding.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
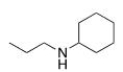
A) N-propylhexanamine
B) N-propylaniline
C) N-ethylcyclohexylamine
D) N-propylcyclohexanamine
F) None of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the name given to naturally occurring amines derived from plant sources?
A) Enamines
B) Imines
C) Alkaloids
D) Alkamines
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major organic product of the following reaction. 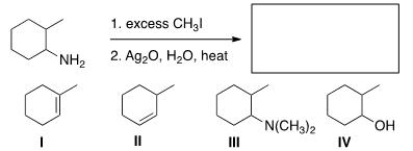
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product of the following reaction. 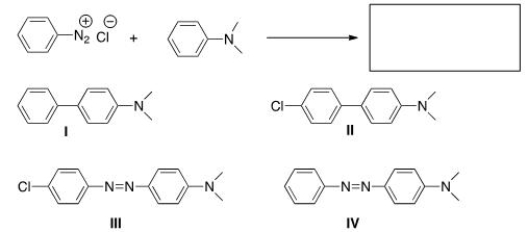
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides cannot be used to prepare primary amines by the Gabriel synthesis?
A) 2-Bromo-2-methylbutane
B) 1-Bromo-2-methylbutane
C) 2-Bromo-3-methylbutane
D) 1-Bromo-3-methylbutane
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product of the following reaction. 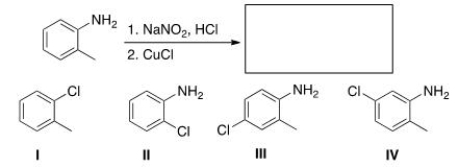
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are 1°, 2°, and 3° alkylamines more basic than ammonia (NH3) ?
A) Because of the electron-withdrawing inductive effect of the alkyl groups
B) Because of the steric hindrance of the alkyl groups
C) Because of the resonance delocalization of the alkyl groups
D) Because of electron-donating inductive effect of the alkyl groups
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following amines in order of decreasing water solubility, putting the most soluble amine first. 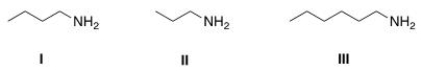
A) I > II > III
B) II > I > III
C) III > I > II
D) II > III > I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate bond angle of the substituents around a nitrogen atom in amines?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following amines are classified as primary (1°) amines?
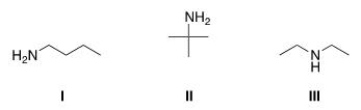
A) I
B) II
C) III
D) I and II
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 53
Related Exams