A) I = Linear; II = tetrahedral; III = trigonal planar; IV = tetrahedral
B) I = Linear; II = tetrahedral; III = trigonal planar; IV = linear
C) I = Trigonal planar; II = linear; III = tetrahedral; IV = trigonal planar
D) I = Tetrahedral; II = trigonal planar; III = linear; IV = tetrahedral
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are around phosphorus in phosphoric acid (H3PO4) ?
A) 6
B) 8
C) 10
D) 12
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the C-H s bonding molecular orbitals of ethane,CH3CH3?
A) Csp2 + H1s
B) Csp3 + H1s
C) C2p + H1s
D) Csp + H1s
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the C-H s bonding molecular orbitals of acetylene,C2H2?
A) Csp + H1s
B) C2p +H1s
C) Csp3 + H1s
D) Csp2 + H1s
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate bond angle for the C-C-N bond in acetonitrile,CH3CN?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has the greatest difference in electronegativity (DE) between the two different elements?
A) CO2
B) H2S
C) NH3
D) H2O
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the nitrogen atom in the ammonium cation,NH4+?
A) sp3
B) sp2
C) sp
D) p
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules are constitutional isomers? 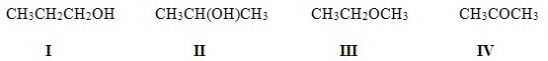
A) I,II,IV
B) II,III,IV
C) I,III,IV
D) I,II,III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pair does not represent resonance structures? 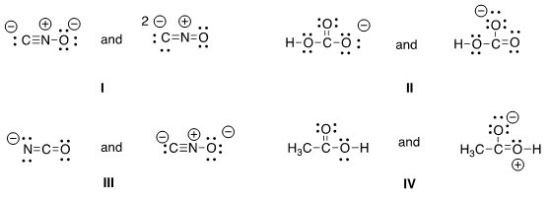
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a resonance structure of the compound below? 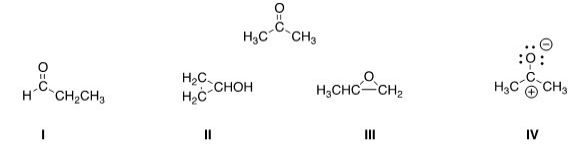
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C2H6O?
A) 1
B) 2
C) 3
D) 4
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of the condensed structure,CH3COCH3,to a Lewis structure? 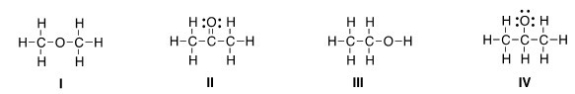
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry around the boron atom in BH3?
A) Tetrahedral
B) Trigonal Planar
C) Trigonal Pyramidal
D) Linear
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When forming molecular orbitals from atomic orbitals,what is the order of increasing C-H bond strength for the following set? 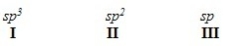
A) II < I < III
B) III < I < II
C) III < II < I
D) I < II < III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the order of decreasing bond length for a C-C bond composed of the following molecular orbitals? 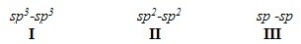
A) I > III > II
B) I > II > III
C) III > II > I
D) II > III > I
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which structure is the hybridization incorrect? 
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would most likely form an ionic bond? 
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain both covalent and ionic bonds? 
A) I,II
B) I,IV
C) II,III
D) II,IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has non-polar covalent bonds?
A) CO2
B) N2
C) CCl4
D) HF
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate value of the H-C-H bond angle in methane,CH4?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 77
Related Exams